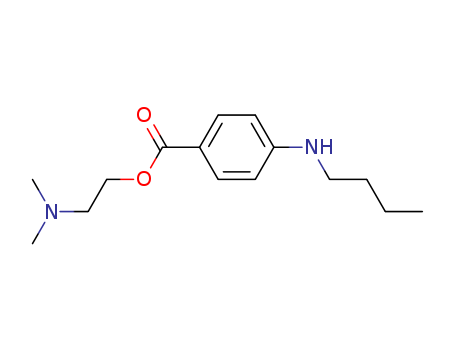
Product Details
94-24-6 Properties
- Molecular Formula:C15H24N2O2
- Molecular Weight:264.368
- Appearance/Colour:White crystalline powder
- Vapor Pressure:2.87E-06mmHg at 25°C
- Melting Point:41.0 to 45.0 °C
- Refractive Index:1.537
- Boiling Point:389.4 °C at 760 mmHg
- PKA:pKa 8.33±0.03(H2O t = 20.0 I = 0.10 (KCl)) (Uncertain)
- Flash Point:189.3 °C
- PSA:41.57000
- Density:1.044 g/cm3
- LogP:2.69000
94-24-6 Usage
Uses
Tetracaine is used in assessing the potential for drug-nanoparticle surface interactions to improve drug penetration into the skin.
Description
Tetracaine is a local anesthetic or numbing medicine that is used to numb the throat, eyes, or nose. Normally, the drug is administered before starting a surgery to decrease pain from the procedure. Tetracaine is applied to the eyes 30 minutes before the start of an intravenous and its effects can last up to 15 minutes.
Mechanism of Action
Tetracaine acts by blocking the nerve systems. It does this by blocking the sodium ion channels needed for the conduction and initiation of neural impulses, thereby affecting the local anesthesia. Tetracaine acts by altering the function of calcium release channels that controls the release of calcium frim intracellular cells.
Precautions
No studies have revealed whether the medicine can affect an unborn child; however, it is important to inform the doctor if pregnant while using it. Informing the doctor is paramount if breastfeeding a baby, as no known research has illustrated whether tetracaine can pass into breast milk or harm a nursing baby. To make tetracaine safe, it is important to inform the doctor if one has a history of spinal or brain injury, heart diseases, eye problems, a blood vessel disorder, open sores, skin injury in the area where the medicine will be applied. Tetracaine should never be orally taken since it is typically designed for use on the skin only. If the medicine gets in the mouth, nose, vagina, or rectum, rinse with water.
Application
Tetracaine topical (for the skin) is used to numb various body parts before a surgical procedure or a medical test. Conversely, tetracaine ophthalmic drops are used as local anesthetic for the eyes.
Dosage
Always follow all direction on the prescription label. Do not use the medicine in smaller or larger amounts or for longer than recommended.
Side Effects
Seek emergency medical advice in case of any of the following allergic reactions: difficult breathing, hives, swelling of the throat, tongue, lips, or face. Stop using the drug and call the physician if one has severe burning, swelling, stinging, or other irritation of the treated skin. Other alarming symptoms include eye watering, irritation, and increased sensitivity to light. Common side effects include burning, stinging, or itching at the point of application of the medicine; numbness in places where tetracaine has been accidentally applied or skin redness or tenderness. Nonclinical Toxicology Studies conducted to reveal the genotoxicity of tetracaine have not indicated its mutagenesis, carcinogenicity, or impairment of fertility.
Chemical Properties
White or light-yellow, waxy solid. Very slightly soluble in water; soluble in alcohol, ether, benzene, chloroform.
Uses
analgesic
Uses
Tetracaine has been used as an anesthetic and to lower the glucose synthesis in upper small intestinal infusions. It has also been used to inhibit ryanodine receptors (RyRs) in rats.
Uses
Topical ophthalmic anesthetic; used for spinal anesthesia
Uses
Tetracaine, also known as amethocaine, is the most potent and lipidsoluble ester local anaesthetic and has significant potential for toxicity if used systemically. It is commonly used as a 4% gel for topical application to decrease the pain from venous puncture or cannulation.
Definition
ChEBI: A benzoate ester in which 4-N-butylbenzoic acid and 2-(dimethylamino)ethanol have combined to form the ester bond; a local ester anaesthetic (ester caine) used for surface and spinal anaesthesia.
Biological Functions
Tetracaine hydrochloride (Pontocaine) is an ester of PABA that is an effective topical local anesthetic agent and also is quite commonly used for spinal (subarachnoid) anesthesia. Epinephrine is frequently added to prolong the anesthesia.Tetracaine is considerably more potent and more toxic than procaine and cocaine. It has approximately a 5-minute onset and 2 to 3 hours of action.
General Description
Tetracaine was developed to address the low potency andshort duration of action of procaine and chloroprocaine. The addition of the butyl side chain on the para nitrogen increasesthe lipid solubility of the drug and enhances the topical potencyof tetracaine. The plasma half-life is 120 to 150 seconds.Topically applied tetracaine to unbroken skin requires 30 to 45minutes to confer topical anesthesia. Tetracaine 4% gel is superiorthan eutectic mixture with lidocaine (EMLA) (an emulsionof lidocaine and prilocaine) in preventing pain associatedwith needle procedures in children. Tetracaine metabolism issimilar to procaine ester metabolism yielding parabutylaminobenzoicacid and dimethylaminoethanol and conjugatesexcreted in the urine. The pKa of the dimethylated nitrogen is8.4 and tetracaine is formulated as a hydrochloride salt with apH of 3.5 to 6.0. The increased absorption from topical siteshas resulted in reported toxicity. Overdoses of tetracaine mayproduce central nervous system (CNS) toxicity and seizure activitywith fatalities from circulatory depression reported.No selective cardiac toxicity is seen with tetracaine althoughhypotension has been reported. Tetracaine is employed for infiltrationanesthesia, spinal anesthesia, or topical use.
Biochem/physiol Actions
Tetracaine also refers as 4-(Butylamino)benzoic acid 2-(dimethylamino)ethyl ester interfere with calcium movement in muscle and non-muscle cells and can inhibit potassium-induced as well as caffeine-induced shortening of outer hair cells (OHC′s). But, the product can′t inhibit electrically-induced shortening of OHC′s.The product can also stimulate caspase activation and inhibits pro-survival signalling pathways which in turn induce human renal cell apoptosis.
Veterinary Drugs and Treatments
Tetracaine is more irritating than proparacaine but is sometimes used in veterinary medicine. It is indicated to produce local anesthesia of short duration for ophthalmic procedures including measurement of intraocular pressure (tonometry), removal of foreign bodies and sutures, and conjunctival and corneal scraping in diagnosis and gonioscopy. Tetracaine is also indicated to produce local anesthesia prior to surgical procedures in humans such as cataract extraction and pterygium excision, usually as an adjunct to locally injected anesthetics. Ophthalmic solutions used for intraocular procedures should be preservative-free. Preservatives may cause damage to the corneal epithelium if a significant quantity of solution enters the eye through the incision.
Uses
Tetracaine is used in assessing the potential for drug-nanoparticle surface interactions to improve drug penetration into the skin.
Description
Tetracaine is a local anesthetic or numbing medicine that is used to numb the throat, eyes, or nose. Normally, the drug is administered before starting a surgery to decrease pain from the procedure. Tetracaine is applied to the eyes 30 minutes before the start of an intravenous and its effects can last up to 15 minutes.
Mechanism of Action
Tetracaine acts by blocking the nerve systems. It does this by blocking the sodium ion channels needed for the conduction and initiation of neural impulses, thereby affecting the local anesthesia. Tetracaine acts by altering the function of calcium release channels that controls the release of calcium frim intracellular cells.
Precautions
No studies have revealed whether the medicine can affect an unborn child; however, it is important to inform the doctor if pregnant while using it. Informing the doctor is paramount if breastfeeding a baby, as no known research has illustrated whether tetracaine can pass into breast milk or harm a nursing baby. To make tetracaine safe, it is important to inform the doctor if one has a history of spinal or brain injury, heart diseases, eye problems, a blood vessel disorder, open sores, skin injury in the area where the medicine will be applied. Tetracaine should never be orally taken since it is typically designed for use on the skin only. If the medicine gets in the mouth, nose, vagina, or rectum, rinse with water.
Application
Tetracaine topical (for the skin) is used to numb various body parts before a surgical procedure or a medical test. Conversely, tetracaine ophthalmic drops are used as local anesthetic for the eyes.
Dosage
Always follow all direction on the prescription label. Do not use the medicine in smaller or larger amounts or for longer than recommended.
Side Effects
Seek emergency medical advice in case of any of the following allergic reactions: difficult breathing, hives, swelling of the throat, tongue, lips, or face. Stop using the drug and call the physician if one has severe burning, swelling, stinging, or other irritation of the treated skin. Other alarming symptoms include eye watering, irritation, and increased sensitivity to light. Common side effects include burning, stinging, or itching at the point of application of the medicine; numbness in places where tetracaine has been accidentally applied or skin redness or tenderness. Nonclinical Toxicology Studies conducted to reveal the genotoxicity of tetracaine have not indicated its mutagenesis, carcinogenicity, or impairment of fertility.
Chemical Properties
White or light-yellow, waxy solid. Very slightly soluble in water; soluble in alcohol, ether, benzene, chloroform.
Uses
analgesic
Uses
Tetracaine has been used as an anesthetic and to lower the glucose synthesis in upper small intestinal infusions. It has also been used to inhibit ryanodine receptors (RyRs) in rats.
Uses
Topical ophthalmic anesthetic; used for spinal anesthesia
Uses
Tetracaine, also known as amethocaine, is the most potent and lipidsoluble ester local anaesthetic and has significant potential for toxicity if used systemically. It is commonly used as a 4% gel for topical application to decrease the pain from venous puncture or cannulation.
Definition
ChEBI: A benzoate ester in which 4-N-butylbenzoic acid and 2-(dimethylamino)ethanol have combined to form the ester bond; a local ester anaesthetic (ester caine) used for surface and spinal anaesthesia.
Biological Functions
Tetracaine hydrochloride (Pontocaine) is an ester of PABA that is an effective topical local anesthetic agent and also is quite commonly used for spinal (subarachnoid) anesthesia. Epinephrine is frequently added to prolong the anesthesia.Tetracaine is considerably more potent and more toxic than procaine and cocaine. It has approximately a 5-minute onset and 2 to 3 hours of action.
General Description
Tetracaine was developed to address the low potency andshort duration of action of procaine and chloroprocaine. The addition of the butyl side chain on the para nitrogen increasesthe lipid solubility of the drug and enhances the topical potencyof tetracaine. The plasma half-life is 120 to 150 seconds.Topically applied tetracaine to unbroken skin requires 30 to 45minutes to confer topical anesthesia. Tetracaine 4% gel is superiorthan eutectic mixture with lidocaine (EMLA) (an emulsionof lidocaine and prilocaine) in preventing pain associatedwith needle procedures in children. Tetracaine metabolism issimilar to procaine ester metabolism yielding parabutylaminobenzoicacid and dimethylaminoethanol and conjugatesexcreted in the urine. The pKa of the dimethylated nitrogen is8.4 and tetracaine is formulated as a hydrochloride salt with apH of 3.5 to 6.0. The increased absorption from topical siteshas resulted in reported toxicity. Overdoses of tetracaine mayproduce central nervous system (CNS) toxicity and seizure activitywith fatalities from circulatory depression reported.No selective cardiac toxicity is seen with tetracaine althoughhypotension has been reported. Tetracaine is employed for infiltrationanesthesia, spinal anesthesia, or topical use.
Biochem/physiol Actions
Tetracaine also refers as 4-(Butylamino)benzoic acid 2-(dimethylamino)ethyl ester interfere with calcium movement in muscle and non-muscle cells and can inhibit potassium-induced as well as caffeine-induced shortening of outer hair cells (OHC′s). But, the product can′t inhibit electrically-induced shortening of OHC′s.The product can also stimulate caspase activation and inhibits pro-survival signalling pathways which in turn induce human renal cell apoptosis.
Veterinary Drugs and Treatments
Tetracaine is more irritating than proparacaine but is sometimes used in veterinary medicine. It is indicated to produce local anesthesia of short duration for ophthalmic procedures including measurement of intraocular pressure (tonometry), removal of foreign bodies and sutures, and conjunctival and corneal scraping in diagnosis and gonioscopy. Tetracaine is also indicated to produce local anesthesia prior to surgical procedures in humans such as cataract extraction and pterygium excision, usually as an adjunct to locally injected anesthetics. Ophthalmic solutions used for intraocular procedures should be preservative-free. Preservatives may cause damage to the corneal epithelium if a significant quantity of solution enters the eye through the incision.
InChI:InChI=1/C15H24N2O2/c1-4-5-10-16-14-8-6-13(7-9-14)15(18)19-12-11-17(2)3/h6-9,16H,4-5,10-12H2,1-3H3
94-24-6 Relevant articles
A new method of dicaine synthesis from p-nitrobenzoic acid ethylate
Abdullaev
, p. 28 - 31 (2002)
-
Visible Light-Mediated (Hetero)aryl Amination Using Ni(II) Salts and Photoredox Catalysis in Flow: A Synthesis of Tetracaine
Park, Boyoung Y.,Pirnot, Michael T.,Buchwald, Stephen L.
, p. 3234 - 3244 (2020/02/04)
We report a visible light-mediated flow process for C-N cross-coupling of (hetero)aryl halides with a variety of amine coupling partners through the use of a photoredox/nickel dual catalyst system. Compared to the method in batch, this flow process enables a broader substrate scope, including less-activated (hetero)aryl bromides and electron-deficient (hetero)aryl chlorides, and significantly reduced reaction times (10 to 100 min). Furthermore, scale up of the reaction, demonstrated through the synthesis of tetracaine, is easily achieved, delivering the C-N cross-coupled products in consistently high yield of 84% on up to a 10 mmol scale.
Preparation method of tetracaine hydrochloride
-
Paragraph 0014, (2017/04/22)
The invention relates to the technical field of preparation method of tetracaine hydrochloride. The preparation method comprises the preparation steps: carrying out a reaction of p-nitrobenzoyl chloride (2) and 2-dimethylamino-1-ethanol (3) to generate p-nitrobenzoic acid-2-dimethylamino ethyl (4), reducing the compound (3) to obtain p-aminobenzoic acid-2-dimethylamino ethyl (4), generating pontocaine (7) from a compound (5) and 1-bromobutane (6) under alkaline conditions, and finally carrying out a reaction of the pontocaine (7) with HCl to generate tetracaine hydrochloride (1).
Block of cyclic nucleotide-gated channels by tetracaine derivatives: Role of apolar interactions at two distinct locations
Strassmaier, Timothy,Kirk, Sarah R.,Banerji, Tapasree,Karpen, Jeffrey W.
, p. 645 - 649 (2008/09/19)
A series of new tetracaine derivatives was synthesized to explore the effects of hydrophobic character on blockade of cyclic nucleotide-gated (CNG) channels. Increasing the hydrophobicity at either of two positions on the tetracaine scaffold, the tertiary amine or the butyl tail, yields blockers with increased potency. However, shape also plays an important role. While gradual increases in length of the butyl tail lead to increased potency, substitution of the butyl tail with branched alkyl or cyclic groups is deleterious.
94-24-6 Process route
-
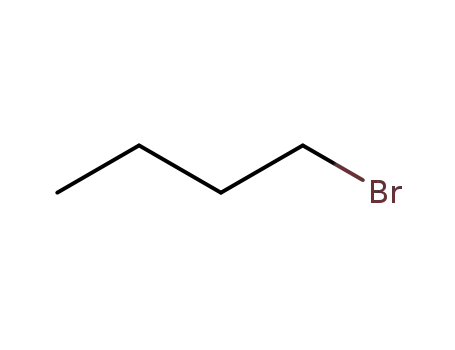
-
109-65-9
1-bromo-butane

-
-
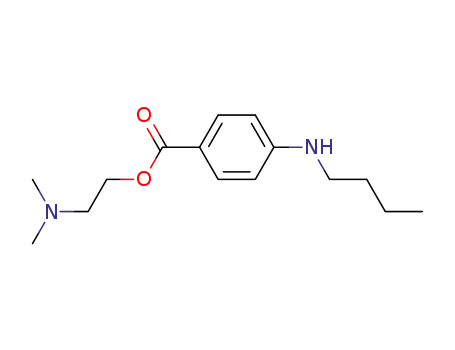
10012-47-2
Dimethylprocaine

-
-
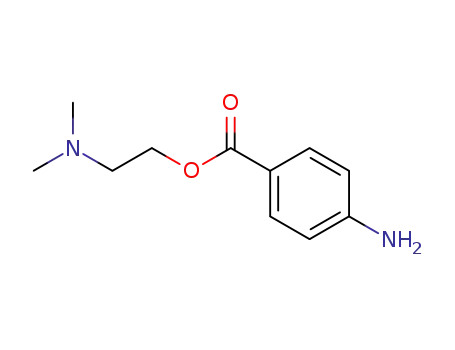
94-24-6
amethocaine
| Conditions | Yield |
|---|---|
|
With
potassium carbonate;
In
N,N-dimethyl-formamide;
at 50 ℃;
for 5h;
|
82.8% |
|
With
butan-1-ol;
|
-
-
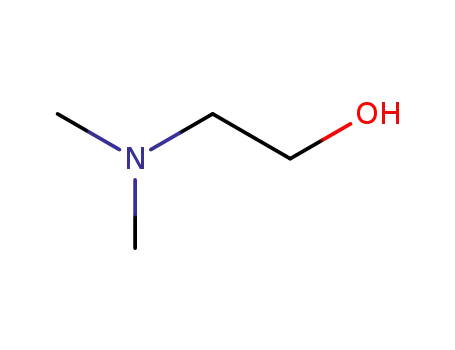
108-01-0
2-(N,N-dimethylamino)ethanol

-
-
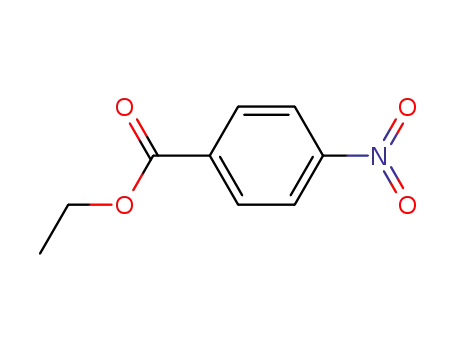
99-77-4
ethyl 4-nitrobenzoate

-
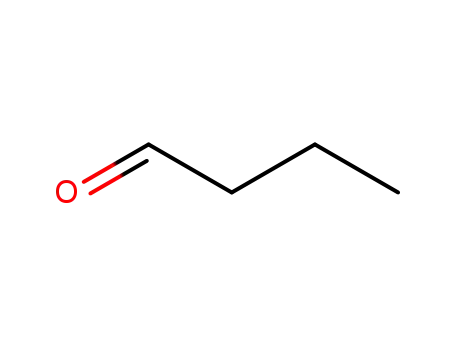
-
123-72-8
butyraldehyde

-

-
94-24-6
amethocaine
| Conditions | Yield |
|---|---|
|
With
hydrogen;
palladium-containing anion exchanger AV-17-8;
In
ethanol;
at 45 ℃;
under 760 Torr;
Further Variations:;
Catalysts;
concentrations;
Kinetics;
|
94% |
|
With
hydrogen;
palladium-containing anion exchanger AV-17-8;
In
ethanol;
at 45 ℃;
under 760 Torr;
|
94% |
94-24-6 Upstream products
-
108-01-0

2-(N,N-dimethylamino)ethanol
-
4740-24-3
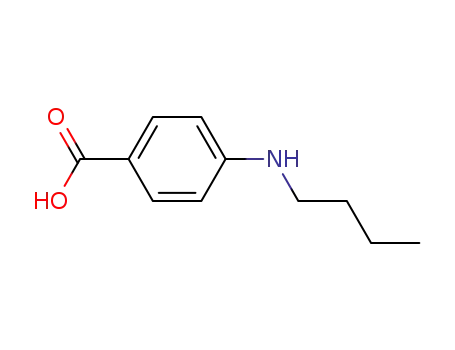
4-(N-butylamino)benzoic acid
-
38152-22-6
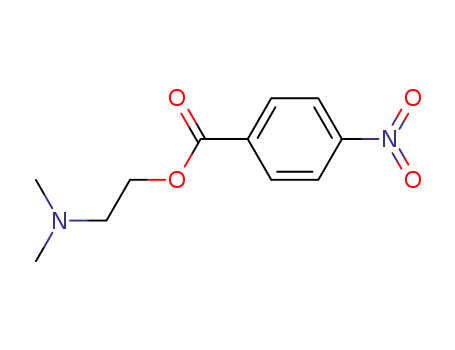
4-nitrobenzoic acid 2-(dimethylamino)ethyl ester
-
122-04-3
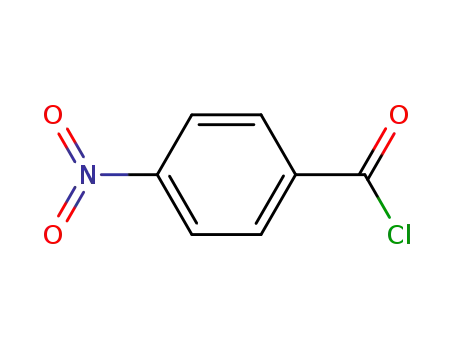
4-nitro-benzoyl chloride
94-24-6 Downstream products
-
55750-02-2
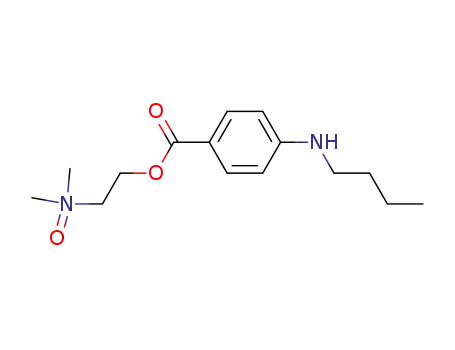
4-butylamino-benzoic acid-[2-(dimethyl-oxy-amino)-ethyl ester]
-
136-47-0
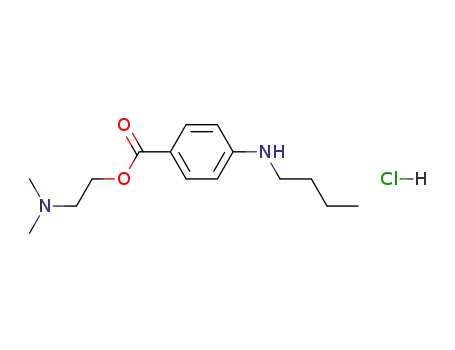
tetracaine hydrochloride